The new method will be applicable to atomic-resolution imaging of complex biomolecules when even more powerful x-ray lasers, currently under construction, are available. The technique will allow scientists to gain insight into the fields of materials science, plasma physics, biology and medicine.
The scientists, part of an international collaboration led by Lawrence Livermore National Laboratory's (LLNL) Henry Chapman and Janos Hajdu of Uppsala University in Sweden, achieved the feat using the world's first soft x-ray free-electron laser, located at the FLASH facility at Deutsches Elektronen-Synchrotron (DESY) in Hamburg. Their work will appear on the cover of the December issue of the journal Nature Physics (12 November 2006 | doi:10.1038/nphys461).
The experiment suggests that in the near future, images from nanoparticles and even large individual macromolecules -- viruses or cells -- may be obtained using a single, ultrashort high-intensity laser pulse before the sample explodes and turns into a plasma. This means that scientists could better understand the structure of macromolecular proteins without crystallizing them, which is required in conventional x-ray structure analysis, and have the ability to rapidly study all classes of proteins.
(From Photonics.com)
2 comments:
科学家创造原子尺度超快成像记录
教育部科技发展中心网2006年11月14日报道 美国Livermore国家实验室(LLNL)的科学家们在11月12日的《Nature Physics》上发表文章,声明他们第一次使用超短超强的X射线脉冲对蛋白质等目标进行原子尺度的无损成像,并取得了25飞秒的快速成像记录。
应用这种方法和正在建造的更强的X射箭激光器,人们将能够对复杂的生物分子进行原子分辨的成像。科学家们将对材料科学、等离子体物理、生物医学等领域有更深刻的理解。
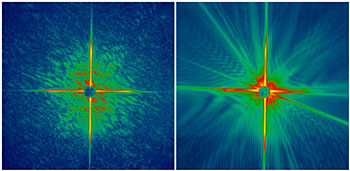
Livermore的科学家使用位于德国汉堡的德国电子同步加速器(DESY)记录了在X射线激光破坏被测物前所成的纳米尺度的衍射图形,然后根据衍射图形使用他们自己发明的算法来重建出被测物的图像。这种成像技术无需高分辨率的透镜组,能够应用于原子尺度分辨的成像。这种成像技术的空间分辨率为50纳米,比光学显微镜要高10倍。
之前的理论指出高分子、病毒或细胞在被超快超强的X射线脉冲破坏成等离子体之前,其衍射图形是可能被记录下来的。这意味着科学家们无需使其成型就能够了解多种蛋白质高分子的结构。四种模型的计算机模拟的结果都支持了这种理论。但是在此之前,这一理论还从来没有实验验证过。
Livermore的科学家们首先提出了验证这一理论的实验,这个实验在DESY的FLASH设备完成。FLASH由自发辐射的自放大原理产生了软X射线自由电子激光(FEL)脉冲。这一脉冲比之前世界上最亮的X射线源要亮一千万倍。在实验中,脉冲捕获图像仅使用了25飞秒。
《自然—物理学》 X射线成像创新纪录
拍摄一个微米尺度大小的棍棒形态的X射线图像只用了25飞秒的时间,这项破纪录的工作是由Henry Chapman和同事完成的,这一研究成果发表在12月号的《自然—物理学》上。新技术比传统闪光照相的速度快了约10万亿倍,X射线图像铭刻在以 60000摄氏度高温蒸发而成的金属薄膜上。
这些图像是利用自由电子激光产生的辐射而形成的,这些自由电子激光是今年初在德国的电子同步辐射装置DESY上开始运行的。这个漂亮的技艺是对一种新技术的重要原理求证,从而使得利用传统的同步加速器光源对分子般大小的物质的原子结构进行成像成为可能。
从结构生物学到纳米技术领域,自由电子激光代表了一个激动人心的发展。这些激光产生出强烈、超短的X射线爆,从而可以测量单个有机分子的结构,而不再像传统的X射线分析时需要将分子晶体化。尽管新工作中还没有展示原子尺度的分辨率,但是,当新一代的能量更强的自由电子光源出现后,这个目标很快就能实现。目前,美国斯坦福线型中心正在建造第四代光源——直线型加速器同调光源LCLS。
Post a Comment